Do you ever wonder why sun exposure causes so many harmful effects to the skin, such as burns, signs of aging, hyperpigmentation, and cancer? While the mechanism of UV damage to the skin is multifactorial, one mechanism is UV-induced free radical activity that damages cells.
There’s no question that the number one way to prevent skin damage from the sun’s harmful UV rays is to avoid sun exposure. However, since this is not always possible, daily use of a broad spectrum sunscreen of at least SPF 30 can help to prevent UV-induced free radical damage to the skin. But did you know that certain antioxidants found in skin care products can provide additional protection against UV damage? Read on to learn how antioxidants work and what the studies have found regarding prevention of UV-induced free radical damage.
How do antioxidants work?
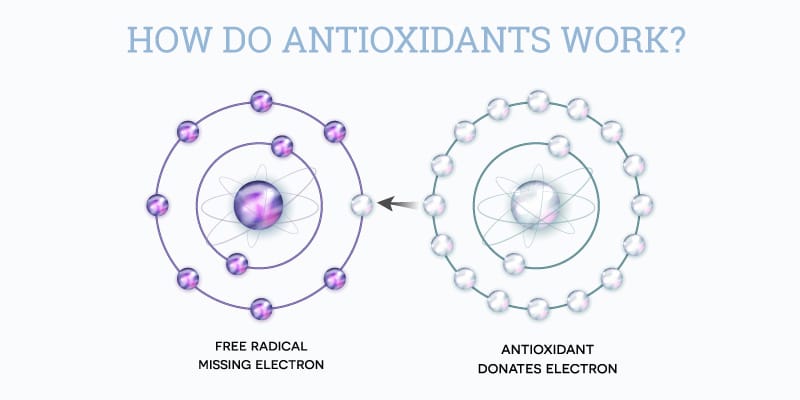
To understand how antioxidants work, it’s first necessary to establish an understanding of free radicals.
Free radicals are atoms that have an unpaired electron in an atomic orbital and are capable of independent existence. (Pharmacogn Rev., 2010) An atomic orbital typically holds two electrons, but during certain reactions a molecule can be left with only one, unpaired, electron in its atomic orbital. An odd number of electrons makes the molecule unstable and highly reactive, resulting in a species that is known as a free radical. The energy of UV radiation penetrating the skin encourages such reactions within cells.
Since free radicals are highly reactive, they want to either donate that single electron to another molecule or accept an electron from another molecule so that they can become stabilized. The “donor” molecule may then become unstable. While a certain level of free radical production is normal, over time free radicals can accumulate and damage cells through vicious chain reactions. The problem arises when the free radicals react with important cellular components, such as DNA, proteins, carbohydrates, or the cell membrane. When this happens, the cells become dysfunctional or undergo apoptosis (cell death).
Fortunately, the body has a defense mechanism against free radicals: antioxidants. Antioxidants work through many different mechanisms, with the main objective being to interfere with oxidative processes that produce harmful free radicals. For example, they may act by scavenging free radicals and “breaking the chain” of reactions, by binding metal ions, or by removing oxidatively damaged biomolecules. (Karger, 2001)
Unfortunately, the body does not produce or ingest enough antioxidants to neutralize all of the free radicals, which leads to oxidative stress. Essentially, oxidative stress is an imbalance between the production of free radicals and the ability of the body to counteract or detoxify their harmful effects through neutralization by antioxidants.
How do free radicals damage the skin?
Free radicals damage the cells of the skin through oxidative stress caused by ultraviolet radiation from sunlight as well as tanning beds. UV radiation causes the production of free radicals, and damage occurs when the critical balance between free radical generation and antioxidant defenses is unfavorable.
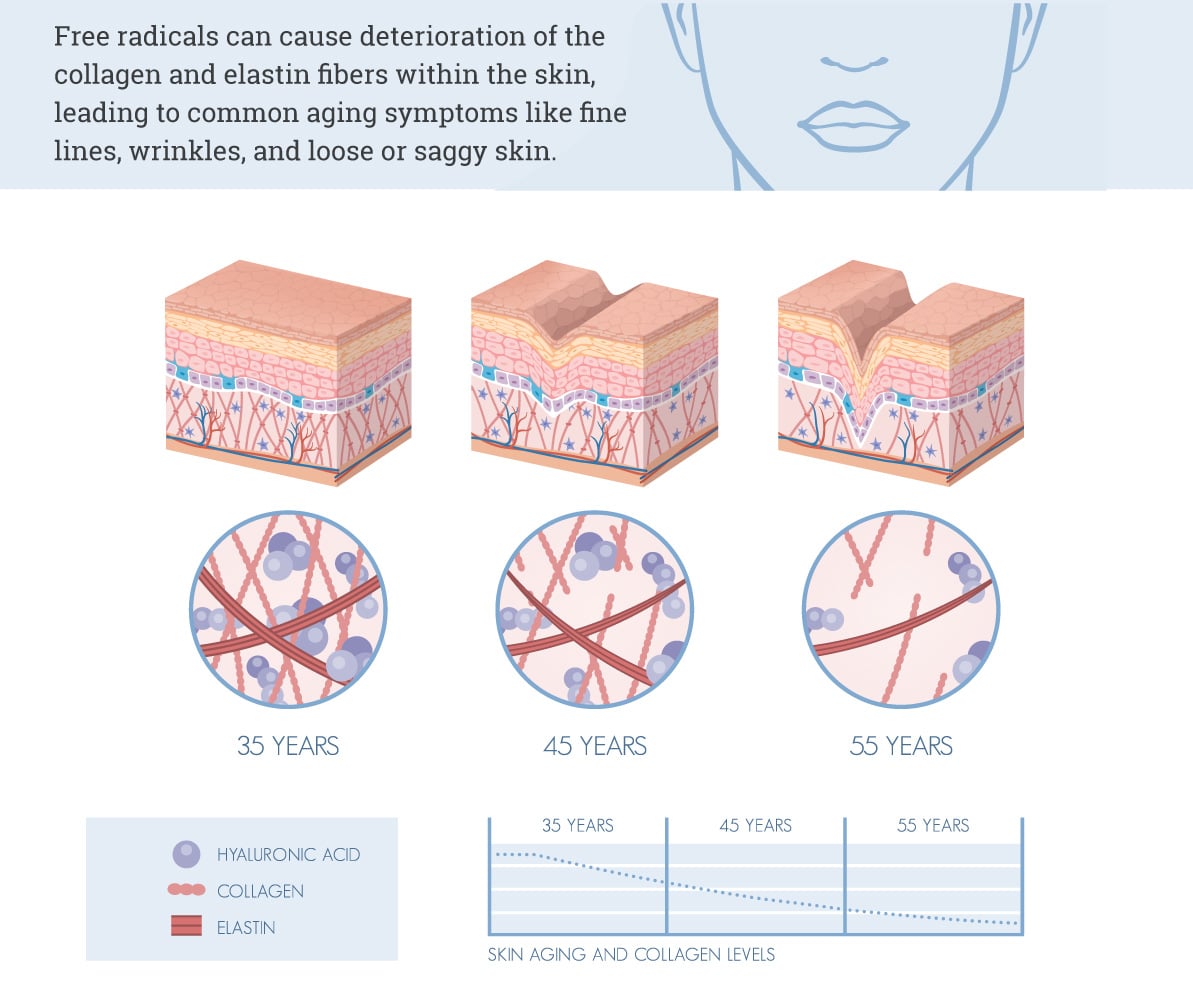
Short-term oxidative stress from UV radiation causes injury to the skin in the form of redness (erythema) and discoloration (hyperpigmentation). However, prolonged sun exposure leads to cumulative damage, which can have more serious health risks such as the development of skin cancer. Additionally, free radicals can cause deterioration of the collagen and elastin fibers within the skin, leading to common aging symptoms like fine lines, wrinkles, and loose or saggy skin.
Which antioxidants can block UV-induced free radical damage?
In her presentation at American Academy of Dermatology’s 2002 annual meeting, Dr. Karen E. Burke, M.D., explained, “There are 3 antioxidants that have been proven to decrease the effect of the sun on the skin and actually prevent further damage: selenium, vitamin E, and vitamin C.” (Medscape) Vitamins C and E are part of “the antioxidant network,” along with glutathione, lipoic acid, and coenzyme Q10. The primary function of the antioxidant network is to work together synergistically to regenerate the other members when they are inactivated through oxidation processes. For example, when vitamin E is used to neutralize a free radical, vitamin C or coenzyme Q10 can donate electrons to vitamin E, effectively “recycling” it in the system. (FutureDerm)
Research suggests that topical application of vitamin C provides protection against damage from UVA radiation. Similarly, vitamin E alone can protect against UVB insult. However, when vitamin C and vitamin E are formulated with a commercial UVA sunscreen (oxybenzone), additional protection against phototoxic damage from both UVA and UVB radiation has been reported. (Acta Derm Venereol. 1996)

Although it is not part of the antioxidant network, selenium, a mineral naturally found in water and certain foods, has been found to have antioxidant activity. A 1985 study by Overvad et al. established that selenium, both topical and oral, could prevent UVB-induced skin tumors in hairless mice. (CED, 2008)
While the presentation by Dr. Burke only identified vitamins C, E, and selenium as having the ability to prevent UV-induced free radical damage, there is also data to suggest that several antioxidants in the polyphenol class offer similar protection.
For example, the natural antioxidant resveratrol, a phytoalexin, is a compound that plants like grapes, berries, and peanuts produce naturally when under attack. Interestingly, research has found that resveratrol can help to prevent UVB-induced damage. A 2008 study published in Photochemistry and Photobiology demonstrated that when human skin cells were treated with resveratrol, the cells’ production of hydrogen peroxide decreased. (Hydrogen peroxide is one type of free radical, known as a reactive oxygen species (ROS), that is produced after UV exposure, leading to cellular damage.)
Another polyphenol antioxidant that has shown efficacy in preventing UV-induced free radical damage is epigallocatechin-3-gallate (EGCG). Don’t let the name fool you, this antioxidant is one that you have heard of before because it can be found in green tea. In a 2001 study in the journal Carcinogenesis, topical application of EGCG to human skin resulted in decreased oxidative stress and an increase in antioxidant enzymes after UV irradiation. Specifically, pretreatment with EGCG was found to restore the UV-induced decrease in the antioxidant glutathione. It also provided protection to the antioxidant enzyme glutathione peroxidase.
To sum it up
Evidence is mounting that antioxidants, specifically selenium, vitamins C and E, and certain polyphenols can help to prevent UV-induced free radical damage. Using products with the appropriate combination and formulation of these antioxidants, along with proper sunscreen application, can add a second layer of protection against the deleterious effects of sun exposure.
Jessica Krant, M.D., MPH, is a board-certified dermatologist with specialized experience in cosmetic, laser, surgical, and medical dermatology, but above all, Dr. Krant is most proud to be a caring, comprehensive physician who takes the time to listen and send her patients home with a smile.













Leave A Comment